
7 diagnoses
2 control groups

3693 donors

43800 original samples

27000 derivative samples









1 male ≤ 65 y.o., no oncological and AD history 2 female undergoing breast plastic surgeries
Cancer is defined as genetic disease. Germline cancer-predisposing mutations are responsible for less then 10% of all cancers, the remaining 90% arise as a result of mutations acquired during lifetime in normal somatic cells. Post-zygotic (or somatic) mutations (PZM) in histologically normal human cells from various organs that develop cancer have increasingly been suggested over the past decade as a major source of cancer driving mutations, but this field is constantly poorly explored.
Biobanking activities in our unit has been carried out from June 2019 in the dispersed form in five cooperating clinical units: Oncology Center in Bydgoszcz; National Institute of Oncology in Cracow; University Clinical Centre in Gdansk, University Hospital in Cracow and Specialist Hospital in Koscierzyna. We designed and implemented dedicated, unique protocols that unified the collection between all the collaborating units. These protocols were developed in close collaboration between the molecularly-oriented team, surgeons involved in patient recruitment and treatment and pathologists collecting samples. The procedures for sample collection meet ethical and legal requirements and were approved by the Independent Bioethics Committee for Research at the Medical University of Gdansk (NKBBN/564/2018).
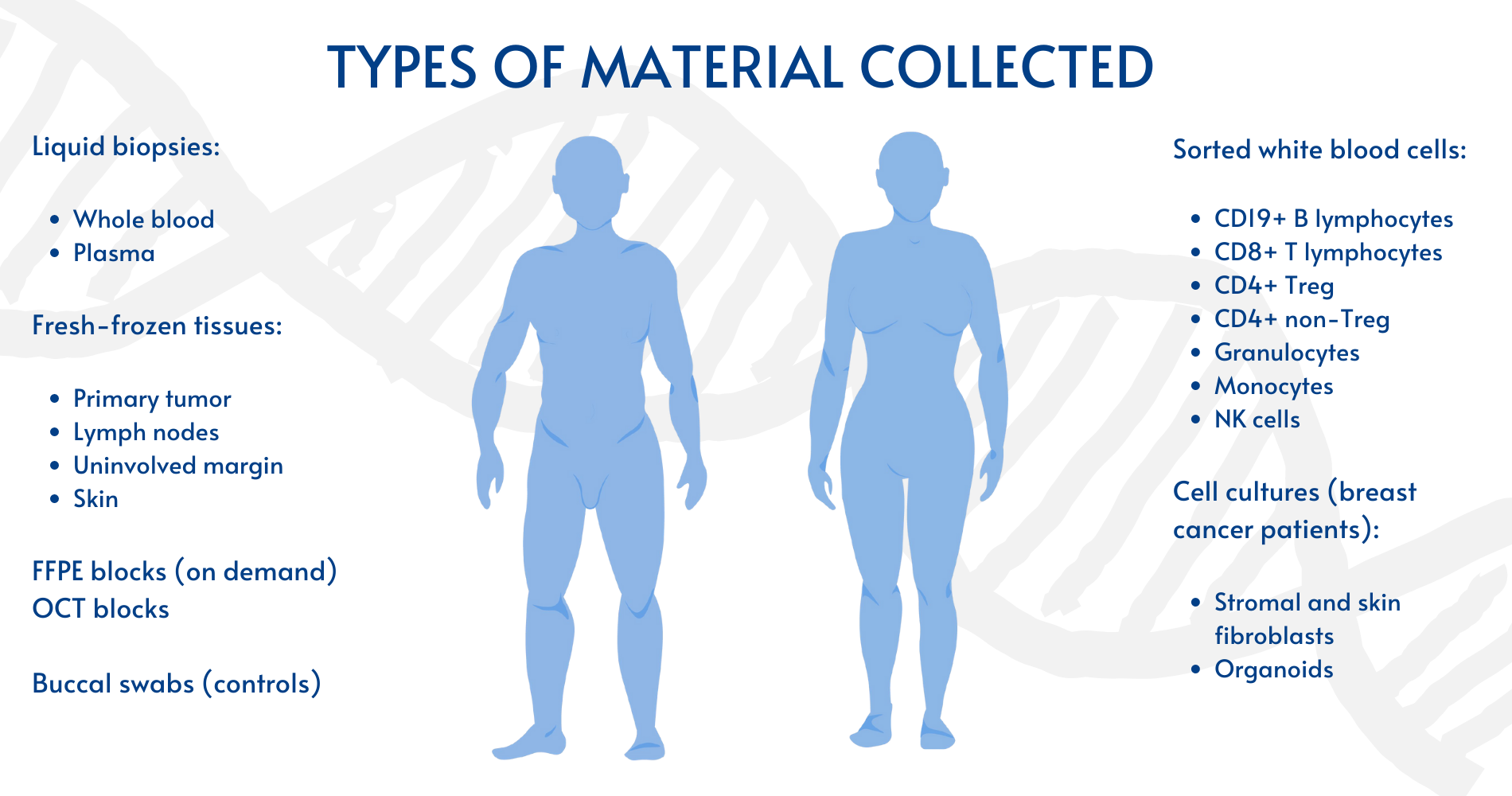
For each oncological donor up to 4 fragments of primary tumor (PT), from 2 to 10 fragments of uninvolved margin (UM), local metastases to lymph nodes (LN) and skin (S) if applicable have been collected and snap frozen. For AD and male control cohort we collect whole blood for sorting leukocytes fractions using FACS and buccal swabs to facilitate Loss of Chromosome Y (LOY) studies. For selected breast cancer patients and female controls we have also initiate primary cultures from skin and uninvolved non-tumorous glandular tissue fragment yielding skin and stromal fibroblasts as well as organoids. Collection of whole blood (up to four 1,5 ml vials) and plasma (2×1,5 ml vials) has been performed for future genetic and proteomic analyses for all cohorts. To assure good quality of RNA/DNA extracted from the collected material, the general condition for all collected samples was a standardized time of maximum 2 hours between tumor/organ resection and the moment of—80-degrees freezing for specimens dissected by the pathologist.
The computerization and semi-automation of the sample collection was implemented from the beginning at each satellite hospital. Together with Bioenit Jakub Szymanowski company we designed, developed and implemented three IT solutions:
- MABData1 – software for registration of patients, clinical data and samples,
- MABData2 – a complete, stand-alone biobank management system,
- Bio3P – cloud product with the potential for commercialization in SaaS model.
The setup of our biobank should allow a wide range of “omics”- and other methods to be applied in studies of the collected clinical material. With the largest oncological collection in Poland we are open for various forms of scientific collaboration. Feel free to contact us: mab@gumed.edu.pl or natalia.filipowicz@gumed.edu.pl
For more info see also the article on our biobanking collection: Filipowicz et al. 2022.
